Download helpful materials to support you throughout KYMRIAH therapy.
How KYMRIAH May Help
Relapsed/refractory (r/r) B-cell acute lymphoblastic leukemia (ALL) treatment, individualized just for you
KYMRIAH® (tisagenlecleucel) was originally studied in a global clinical trial of children and young adults with relapsed or refractory B-cell ALL, with many achieving an early and lasting remission. While many patients have been successfully treated with KYMRIAH, not all patients will respond the same way.
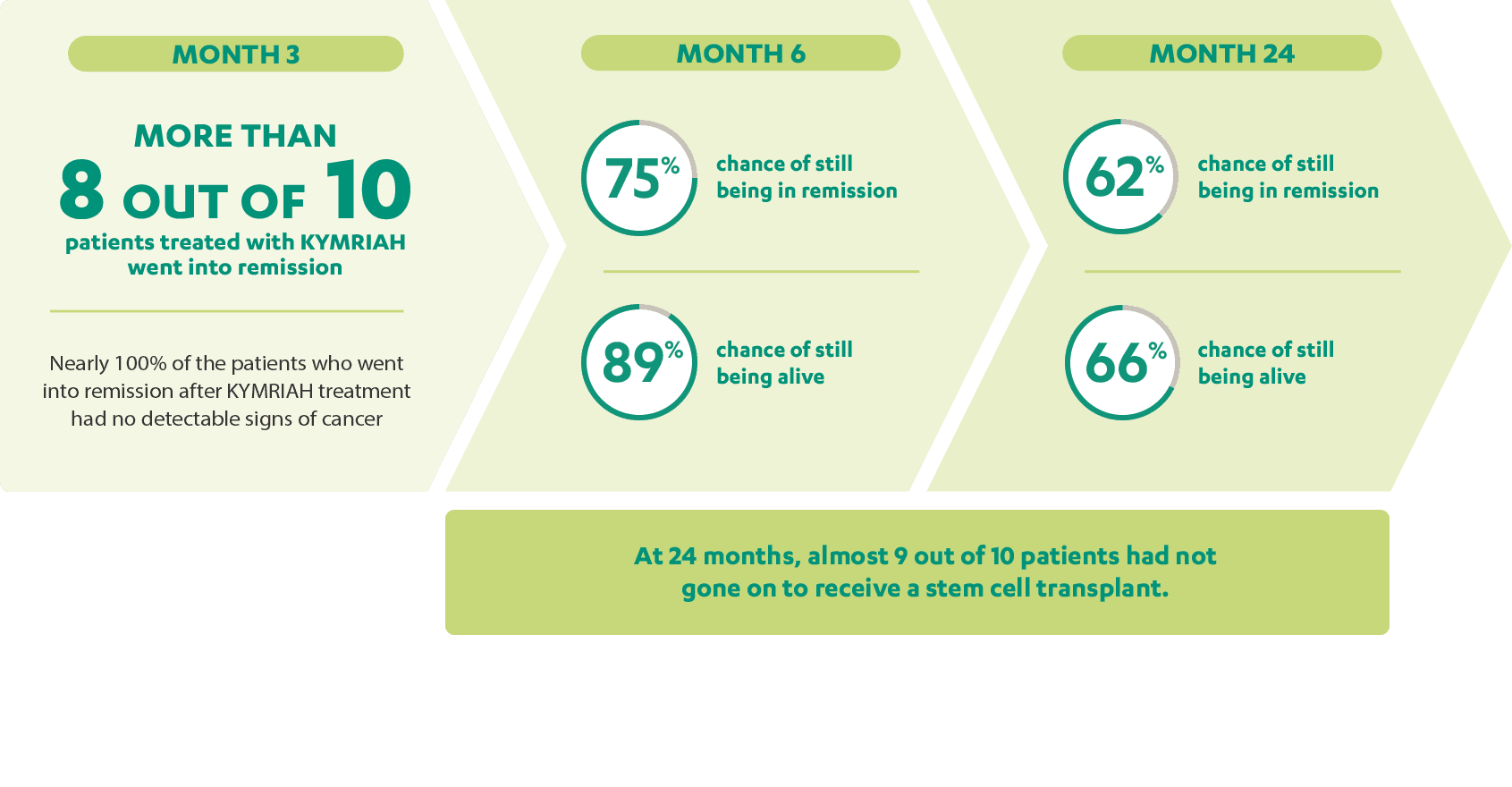
What are the long-term results?
Since KYMRIAH was approved, patients from the clinical trial have been followed to see how well they do over the course of years. A long-term follow-up of patients in the clinical trial was done at 24 months from the date of infusion of KYMRIAH.