Download helpful materials to support you throughout KYMRIAH therapy.
What to Expect With KYMRIAH
What is the process for getting KYMRIAH?
You and your treatment team may be considering KYMRIAH® (tisagenlecleucel). Below is a detailed description of each step so you know what to expect with KYMRIAH therapy. Your treatment team will be there to help guide you through the process. To watch relapsed/refractory (r/r) acute lymphoblastic leukemia (ALL) patients, LJ and Nick, and their families discuss their experience with each step of the KYMRIAH process, click the video clip next to that step.
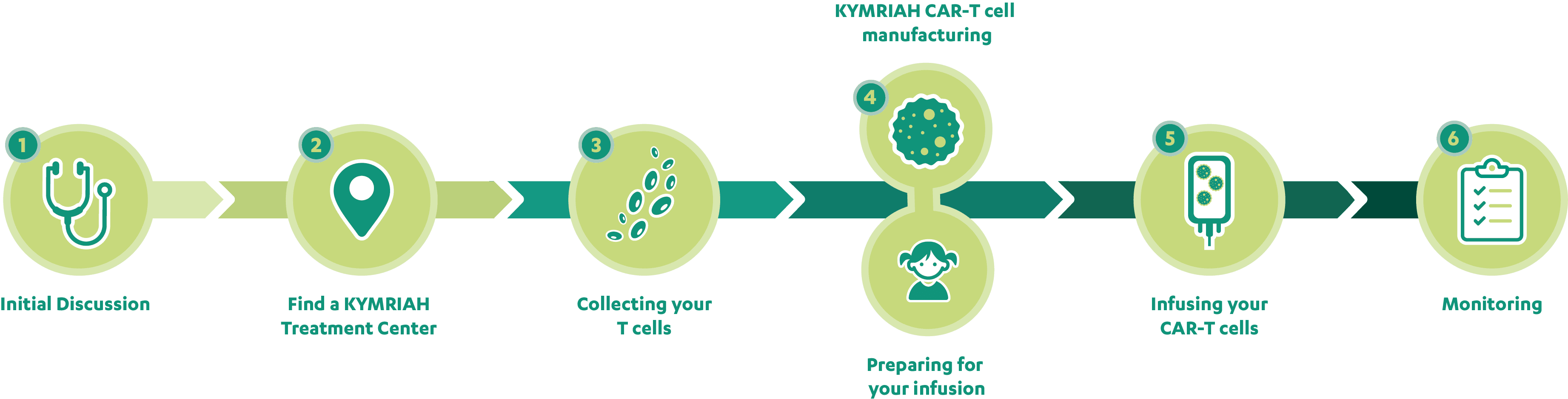
KYMRIAH is a therapy for children and young adults up to 25 years of age with B-cell ALL that is refractory or in second or later relapse. Discuss your treatment history and overall health with your doctor to see if you are a candidate for KYMRIAH therapy. Go to What should I ask my treatment team? for help speaking with your doctor.


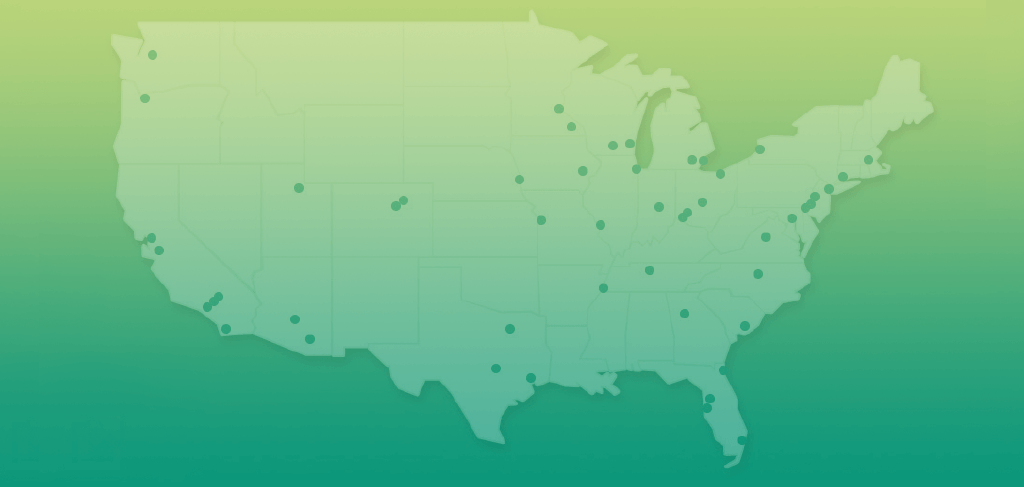
Some people are already receiving treatment for their r/r B-cell ALL at a treatment center that administers KYMRIAH. Others get a referral for KYMRIAH from their primary oncologist. If you haven’t already selected your treatment center, view the list of participating KYMRIAH Treatment Centers, or call KYMRIAH CARES™ at 1-844-4KYMRIAH (1-844-459-6742) for an up-to-date list of treatment centers in the United States so you can find a center near you.
Treatment with KYMRIAH uses your own cells, so the process begins with T cell collection. A small, temporary tube (IV catheter) will be placed into your vein to collect your T cells. This process is called leukapheresis (pronounced "loo-kuh-fuh-REE-sis") and takes about 3 to 6 hours.


Your cells will be sent to a manufacturing facility to be reprogrammed into KYMRIAH CAR-T cells. The process usually takes 3 to 4 weeks, but timing and manufacturing outcomes can vary. About 2 to 11 days before your KYMRIAH CAR-T cells are ready, you may receive a short course of chemotherapy over 2 to 3 days that helps prepare your body to receive your reprogrammed KYMRIAH CAR-T cells. Your treatment team can help guide you through the process. Watch the mechanism of action video to learn more about what happens to your T cells.


Once your KYMRIAH CAR-T cells are ready, they are put back into your body through a standard IV infusion that usually takes less than 30 minutes. About 30 to 60 minutes before your KYMRIAH infusion, you will receive acetaminophen and an antihistamine.
Patients must stay within 2 hours of the treatment center location for at least 4 weeks after being infused.


Once your KYMRIAH CAR-T cells are in your body, they will begin to detect and destroy your cancerous and normal B cells. During the weeks after KYMRIAH infusion, your treatment team will monitor you closely to manage any side effects, some of which may require you to go to the hospital. Remember that some side effects are expected, and your treatment team is prepared to help. Go to What side effects may I experience with KYMRIAH? for more information.


Your treatment team will establish a monitoring plan for ongoing follow-ups. During this period, you will be evaluated to determine if you can get back to your daily activities.
The US Food and Drug Administration (FDA) recommends that all patients who are treated with KYMRIAH be followed for 15 years after infusion. Your treatment team will offer you participation in a long-term registry conducted by the Center for International Blood and Marrow Transplant Research (CIBMTR) for this follow-up. This information is used to help future patients and contributes to the understanding of the effects of CAR-T cell therapy.


The KYMRIAH treatment process is different from that of other cancer therapies. Hear from LJ and Nick's parents to learn how their families managed this process.



You may go see a different doctor who will take some of your cells from your blood and teach them how to look for your advanced blood cancer cells. Once your cells know how to find your advanced blood cancer cells, they will be put back into your body to start fighting your advanced blood cancer.